A project to turn Australia’s low grade iron ore into high grade magnetite at very low temperatures is out of the lab and ready for real world testing, with a government-funded trial proving the concept works.
The idea put to a scientific pressure test is Fortescue’s low‑temperature direct electrochemical reduction (DER) process, which reduces iron ores to iron metal at the super low temperature of 130ºC and doesn’t need hydrogen.
The main green hydrogen options in operation today swap in huge amounts of electricity for coal to power a blast furnace, which works at temperatures around 1000-2000ºC.
With lab- and benchtop-sized electrolysers validating Fortescue’s DER idea, the science is now ready to be tested in a 100 kg-feed pilot that can convert at least half of the feedstock into metallic iron, the final report from the Australian Renewable Energy Agency (ARENA) says.
The process uses a solid-state slurry electrolyser with an alkaline electrolyte designed so they can use intermittent renewable energy to make green iron at low temperatures.
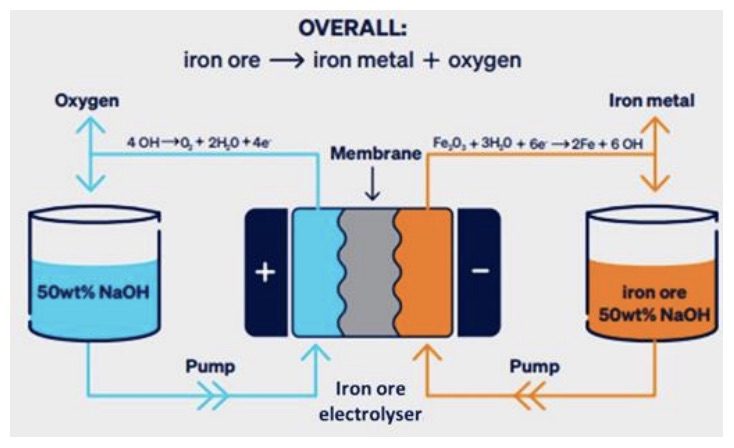
Fortescue founder Andrew Forrest once joked he could tell people about the membrane used in one of the company’s electrochemical iron ore reduction processes, but he’d have to kill them afterwards. The ARENA paper reveals that this DER system uses an AGFA Zirfon membrane.
Green methane or hydrogen can be used in direct reduction technologies, but currently works best on high grade ores as lower grades need extra processing afterwards.
The Fortescue project wants to make ironmaking possible at lower temperatures, with the report saying the technology “has the potential” to deliver energy efficiency in the process and substantially reduce carbon emissions.
What was not part of the report is just how energy intensive the resulting metallic iron will be, with the answer to that question deferred until the pilot.
Value add to low-grade ores
Part of the goal is to lock in the long-term competitiveness of Pilbara iron ore, especially for Fortescue’s lower grade ores which tend to hold 56-59 per cent iron and even now command a lower price than those from rival Pilbara giant Rio Tinto.
Green iron is also one of the green energy threads that Fortescue, owned by billionaire Andrew Forrest, is pulling on to achieve real zero – the removal of all greenhouse gases from a business rather than relying on offsets for the more difficult ones.
The iron ore giant has pulled back from dealing in hydrogen electrolysers, dumping what was supposed to be a flagship green hydrogen facility in Gladstone, Queensland and saying last year it is “refocusing our efforts into the research“.
The company’s hydrogen-based, 1500 kg/year direct reduction iron project at Christmas Creek is still under construction, with operations pencilled in to start this year.
Fortescue has been investigating the low-temperature electrochemical method for some time however.
In 2023, then-Fortescue Future Industries CEO Mark Hutchison bragged that they’d produced 150kg of zero-carbon metallic iron in the lab, with the only clue about the process being a hint about a “membrane” by Forrest.
And in 2024 it launched a huge lawsuit over against a startup founded by three former employees alleging they’d stolen the idea, before it was snuffed out late last year when a Federal Court judge refused to give the company’s lawyers access to all of Element Zero’s work.
So what are these mysterious membranes?
The membranes tested in this study were ceramic–polymer composites, zirconia particles embedded in a matrix of a high heat resistant plastic called polysulfone.
It found the thicker the membrane, the better it was at preventing unwanted particles through but used more energy, and vice versa.
Adding surfactants meant side reactions didn’t happen as often, although that depended on the electrode type and electrolyte.
The ARENA-backed inquiry was to prove up the process for a pilot and position the technology as a low-hydrogen, low temperature alternative, with $5 million in funding in a $43 million project.
And yet the results show that higher operating temperatures still boost production rates and lower energy use, and that the lowest grade of ore, goethite is consumed first, followed by hematite and magnetite.
The inference is that converting goethite to a slightly higher grade of ore before putting it through the electrochemical process will make converting the other grades more efficient.
If you would like to join more than 29,000 others and get the latest clean energy news delivered straight to your inbox, for free, please click here to subscribe to our free daily newsletter.








